We're Making Hand Sanitiser
Times are pretty strange...
When Barry (our chemical wiz) found out a few weeks ago that our local medical clinic couldn't buy hand sanitiser we quickly got to work.
This presented a unique opportunity in trying times. Specifically how to make hand a sanitiser that not only works, but leaves your hands feeling less dry. Barry set about the task of making a hand sanitiser that:
Was easy to spread and apply (to make sure you get good coverage on your hands)
Smelt less like alcohol (even though sanitisers have to be over 60% ethanol)
Could be used on other hard surfaces as a multi-purpose sanitiser
Didn't completely dry out your hands!
He landed on a combination of ethanol and eucalyptus oil (a natural biocide with the added benefit of a pleasant odour!). To help with the spread he added some pharmacy grade propylene glycol.
Luckily we were one of the few chemical companies in Australia that still had a small quantity of ethanol (one of the key ingredients of hand sanitiser) on our factory floor as well as a few other ingredients. In a few days we were able to deliver our first batch and have had really positive feedback since.
We've been working hard to sure up supply and this has meant we're able to reach out to the Aird community just in case people are struggling to find hand sanitiser.
Our Hand and Hard Surface sanitiser is a dangerous good (because of the alcohol) and is a little more liquid rather than a gel (this helps to spread quickly).
Currently we're selling 10L cubes (with a tap and a cap) and 250ml containers with a snap cap.
We're selling this through our stockists so please feel free to reach out to them.
For those unsure of how to purchase please contact us at info@airdchemistry.com and we'll make sure we find a way to get this to you.
We hope that you and your families are staying safe in this socially distant world we live in.
How to Dose and Mix Powers (Cleanskin, Destainex, Oak Restorer)
Dosing and mixing cleaning agents properly can make a big difference to the end result. We’re going to focus on powders and this article will be helpful for Cleanskin, Destainex and Oak Restorer.
PPE
Safety always comes first and it is a good idea to use eye protection for any chemicals you’re mixing. We also recommend gloves as alkaline chemicals dry out your skin.
Safety first - eye protection is a good idea is a must with any chemicals, and gloves look after your hands.
Dosing
The next step is to look at the amount of chemical you need to clean with. Ideally we recommend our products to be used at 1-2% in water. We wanted to make 100L of cleaning solution so this means we need 1kg of Cleanskin.
We suggest weighing out a scoop to 1kg so you it’s easy to dose each time you need to make a batch to clean with.
1kg for 100L of water gives us a 1% solution
Mixing with water
When we mix chemicals we like to have a little bit of water (perhaps 10L) in the drum/bucket that we’re mixing our chemicals in. We then pour the powder in slowly making sure to keep adding water so that the chemical mixes with the water.
Mixing
The idea with mixing is to prevent chemical from sinking to the bottom of the drum and not dissolving. All Aird Chemicals will dissolve in water - the trick here is to pour the chemical in slowly. We like to keep pouring water in throughout as this helps to dissolve the solution. We like to use a stainless steel paddle to mix, however you’re welcome to use any clean plastic or stainless steel implement to give it a good stir.
You’ll know when the solution is ready to go when it goes clear or a milky colour.
Once it is milky like this you’re good to go!
Cleaning Safely With Chemicals
When we set out to develop Cleanskin, we wanted to make a product that was safe. The feedback overwhelmingly from cellar staff was they’d like to use non-dangerous goods. This made perfect sense to us and we know that everyone needs to be vigilant when handling dangerous chemicals.
Here are some of our top tips when working with chemicals.
1. Check the MSDS
Knowledge is power after all. There are risks associated with dangerous goods such as sodium hydroxide or hydrogen peroxide. We see the first step in making sure you’re handling chemicals safely is to know what you’re using. The Material Safety Data Sheet (or Product Safety Data Sheet or Safety Data Sheet) has information about hazardous information and composition of inputs. This can help you identify risks in the work place. All AIRD SDS information is available here.
Anther good resource to check if a chemical is dangerous or not is the Hazardous Chemical Information List.
2. Chemical Storage and Mixing
Some chemicals don’t like others! From basic chemistry we know that acids will react with alkalis (anyone remember the bi-carb soda and vinegar volcanos?). When storing chemicals it is really important that the space is clear, dry with good ventilation and it doesn’t get too hot or cold.
There is a wealth of information on this online – some of our basic recommendations are:
- Keep your acid based products away from alkaline products
- Keep anything with hydrogen peroxide separate from your acids and alkalis (ie Linvasan, PerCitra, Peracetic Acid).
- With liquids make sure that there is adequate bunding (to capture any spills)
- Make sure you have adequate space to utilise and mix chemicals (ie no trip hazards)
- Ensure that labels are visible and easily read
- Make sure pumps have drip catchers
- Know which chemicals can react to heat
3. What are you using the chemical for?
Looking at the application of a chemical is a really important safety consideration. When cleaning it may be possible to use a safer means of achieving the same goal. If it is possible to eliminate dangerous goods from your site then this means that you’ve significantly improved safety.
If it isn’t possible to remove every dangerous good, look at limiting the amount of exposure your staff will have to dangerous goods. Making dangerous goods a last resort throughout the cellar means that staff will be safer.
For example an approach to cleaning could be:
1. Initial water rinse
2. Circulation of non-dangerous chemical
3. Rinse and Review
When staff are at the review stage and further cleaning is needed they can make an educated decision about next steps if they’re required. Most cleaning in the cellar for wine shouldn’t need a dangerous good.
4. Is the product a powder or a liquid?
This may seem trivial however it is important. The risks associated with dangerous goods in a powder form are much higher because the strength of these chemicals are generally much greater. For example, if your winery uses caustic the average powdered form of sodium hydroxide is 99.6% active (caustic pearl), versus the liquid version that is circa 45-50% active. While both are dangerous one is much stronger than the other.
Another risk from powdered caustic is dust. The dust can be inhaled, or can easily get into eyes so it is important that adequate safety equipment is being used. Be very careful disposing with the packaging (ie bags/buckets that chemicals are contained in). Often the packaging that caustic comes in is overlooked by cellar staff and in a powder form it only takes a small amount to do a lot of damage.
Vapours are another risk with powders – especially with caustic. Be very careful as you can get a face full of pH 14 vapours and even with safety equipment this is a high risk.
The downside of dangerous goods as a liquid is that if they spill easily and even at a lower percentage activity level a splash in an eye can be very dangerous.
5. Make sure staff know the WHS plan and are familiar with how to use chemicals and are supervised appropriately
If you don’t already have a WHS plan this is a really useful link. There are lots of resources on the web, and industry trained safety consultants can help make sure that your workplace is safe. When it comes to chemical safety having a WHS plan and making sure that staff are adequately trained is essential.
Working safely is working smarter. In the AIRD range Cleanskin, Destainex, Citsanex, Vinisan, Oak Restorer, Peroxica, Rejuvasol and Wineglass are all non dangerous.
Haven’t tried any of our non-dangerous cleaners and sanitisers lately? Talk to us.
Useful Links:
Cleaning to Organic Standards
Organic wineries often ask us if our products comply with organic standards? Our first question in response is which one! The Department for Agriculture in 2010 outlined that there were in fact 7 bodies that are able to provide organic certification (read more here).
This provides no shortage of confusion! We thought we’d go back to basics and see if we could clarify what organic means and how this impacts hygiene (cleaning and sanitation) product choice.
From the department of Agriculture there were the following organisations that were accredited to provide organic certification:
• AUS-QUAL Limited (AUSQUAL)
• ACO - Australian Certified Organic
• Bio-Dynamic Research Institute (Demeter)
• NASAA - The National Association of Sustainable Agriculture Australia
• OFC - Organic Food Chain
• Safe Food Production Queensland
• Tasmanian Organic Dynamic Producers
(Department of Agriculture Feb 2015)
Organic certifiers all adhere to the minimum requirements set out in the National Standard for Organic and Bio-dynamic produce and may have further requirements on top of this.
So what is organic? From the Australian Government Definition we can define Organic as:
Organic: means the application of practices that emphasise the:
- use of renewable resources; and
- conservation of energy, soil and water; and
- recognition of livestock welfare needs; and
- environmental maintenance and enhancement, while producing optimum quantities of produce without the use of artificial fertiliser or synthetic chemicals.
Furthermore we can define bio-dynamic as follows:
bio-dynamic: means an agricultural system that introduces specific additional requirements to an organic system. These are based on the application of preparations indicated by Rudolf Steiner and subsequent developments for management derived from practical application, experience and research based on these preparations.
Source National Standard for Organic and Bio-Dynamic Produce Edition 3.5 (Feb 2013)
We like what we’re hearing. Using renewable resources, conservation of energy, soil and water and a conscientious approach to the environment. We don’t think any wine maker would disagree with these concepts on the whole.
So the part we are interested in is the chemical part – where chemicals are used for cleaning and sanitation. The good news is that hygiene is factored into producing organic wine – all the accreditation bodies that we’ve worked with and have been able to research, allow a range of chemicals to be used so long as they are followed by a potable water rinse (so that cleaning inputs don’t come into contact with organic produce).
What is concerning is the types of chemicals that are able to be used by wineries undertaking organic production. Our table below gives a summary of some of the inputs that are allowed under different bodies.
Approved organic inputs by accreditation provider
Of course – if you are an organic producer you’ll need to check with your accreditor to verify this.
We think some of these chemicals need review – especially in light of the environmental conservation aims outlined in the National Standards. The reality is that the standard allows halogens (iodine, sodium hypochlorite), quaternary ammonium and salts such as sodium and potassium hydroxide to be used so long as they don’t come into contact with the organic produce.
We see a few issues with this as halogens are toxic to many plants, quaternary ammonium has a large environmental impact (worryingly there are some microbes that are developing a resistance to QAC - Tezel, 2009) and strong alkali salts can lead to sodic soils (you can read more on caustic here).
While we are sure many conscientious wineries won’t be going near some of these products – the fact that they are listed as inputs in national standards is a real environmental risk – completely undermining the environmental principles of what organic produce is about. The potable water rinse – “should” mitigate the risk of using these products. In reality though, mistakes happen. Having the chemical’s like sodium hypochlorite or QAC’s onsite at a busy time of year could mean that the very best of intentions have been mitigated by human error – in doing so contaminating what would otherwise be organic produce.
For organic producers we would always recommend using a bio-degradable detergent. The good news is that AIRD products have always been approved for use in organic facilities. Our products are also low salt, have a low VOC are produced from bio-degradable materials and mostly are non-dangerous (improve safety and environmental impact simultaneously).
And even if you’re not working in an organic facility – your waste water will thank you for using products that biodegrade effectively and have a low environmental impact.
Like to know more? Talk to us.
Further Reading:
ACO - Australian Certified Standard 2013
http://austorganic.com/wp-content/uploads/2013/11/ACOS-2013-final.pdf
Organic Standards and Certification in Australia
Tezel. U, 2009 Fate and effect of Quaternary Ammonium Compounds in Biological Systems, Georgia Institute of Tech
Interestingly from Tezel’s research:
“Given the antimicrobial properties of QACs, microbes that subsist on QACs have to also have developed resistance mechanisms.”
“…strong evidence of biodegradation potential of QACs in biological systems under aerobic, anoxic and anaerobic conditions.”
Cleaning Cross Flow Filters
We're often asked how to clean cross flow filter systems. This can often be a challenge. Cross flow systems remove very fine particles out of wine – and to remove these particles from a filter system can be tricky. The nature of the organic soils – although still wine – often are more gelatin like, and without some thinking before hand it is easy to turn an organic gelatin in the cross flow system into soap!
Often the two key concerns for cleaning are pressure and flow. As organic soils and solids build up in the filter there is an increase in pressure on the pump. While this is building pressure on the pump it is also decreasing the flow rate for the entire machine.
So the cleaning aims seem simple – remove the solids, reduce the pressure and increase the flow. With a few variables in play here (ie the filter type) we have some views on this that might help with cleaning.
If we go back to some basic principles of cleaning in wine the steps are;
- Reduce the surface tension of any solids to wash the soils off the article being cleaned (detergency, surfactants, wetting agents);
- Dissolve (emulsify) any acids/organic soils that can be dissolved.
Once the surface tension of a solid has been reduced (ie that soil can’t hold onto the surface any longer) – it can then be washed off the surface, and if soils can be moved into a solution then they can be washed out of the vessel being cleaned.
This makes sense for tanks – if you can get the soils in the tank into a solution – or get the soils off the wall of the tank you then have a good chance of rinsing out all the organic matter leaving a bright shiny finish.
Where this process comes unstuck for cross flow systems is the type of chemicals that are often used. Many manufacturers recommend caustic to clean with. Our view on caustic is that it doesn’t clean effectively. What we find is that the high pH of caustic products actually increases the surface tension of soils. This means an organic soil is harder to remove from a surface – it is more attached requiring more force to shift it. This is happening chemically in tanks, but also in the cross flow filters.
The compounding issue for cross flow filters is that high pH products on gelatin soils can have the effect of creating soap! The caustic hardens the gelatin, making it extremely difficult to wash from the system.
Another issue we see commonly in bottling lines that can impact cross flow systems is water temperature. If the water used to clean is too hot (ie above 90 degrees C) it will cause a protein char. This stain is burnt on (similar to a chemical burn from caustic) and it is actually caused by just hot water.
The two common problems as we see it, are that current chemicals being used aren’t performing adequately, and that very high water temperatures can be creating a further problem in cleaning cross flow systems too.
For cross flow systems we suggest that moving away from caustic is a good first step to a better clean. A lower pH product, (in our range Cleanskin, Cleanskin K or Oak Restorer) – where the surfactants are food grade, plant based and rinse well, will have a far better impact in reducing surface tension so that soils can be washed from the system. Using warm water (40-60 degrees C) will help with this too.
Cleaning a cross flow system is tricky, it will take effort. In our opinion using quality chemicals and water that isn’t too hot will make the job a lot easier.
All content provided on this blog is for informational purposes only. AIRD Chemistry makes no representations as to the accuracy or completeness of any information on this site or found by following any link on this site. AIRD Chemistry will not be liable for any losses, injuries, or damages from the display or use of this information.
Quality in Chemical Production is in Rapid Decline
The adage that you “get what you pay for” is very true for practically everything, including chemicals. The demand for low cost chemicals has meant manufacturers are either removing or substituting expensive inputs from the end product. The resultant chemical may be masking future problems and is in stark contrast to the direction of the wine industry.
The wine industry has been working hard to change the perception of Australian wines. No doubt, Australia is still making some pretty good low cost wines, however industry bodies are all in agreement that longer term the money is in quality, not quantity.
“…longer term, producers will move towards premium wines, which will bode well for margins and industry profitability.” – Ibis world
We can see this in the sales figures too.
Australian wine sales declined 6% last year, yet the value has increased by 2% (Wine Australia Annual Report).
While no-one will argue that saving costs in wine production is important, longer term making a better quality ‘drop’ is the direction of the industry. However, wineries looking to save on cleaning and sanitation chemicals need to be careful. While wine as an industry in Australia is moving towards a premium price bracket, most chemical manufacturers and suppliers are doing the opposite to secure sales.
There is a significant push towards reducing costs in chemical manufacturing while increasing sales by volume or weight. This is concerning as wineries are facing greater environmental constraints, in order to reduce the amount of chemical being consumed in an effort to be more sustainable.
Saving money in chemical production is pretty simple. Common cost reduction is by reducing material activity via substitution (taking a more expensive compound and replacing with a cheaper input); introducing water soluble fillers (near neutral compounds that have no impact but weight) and lowering the total active ingredients percentage (so more of the product is required to complete the job).
They all have similar themes – the customer will need more chemical to complete the job therefore increasing cost with more purchasing. Another measure chemical producers use to lower costs is to use the same product across multiple industries. Customers should be quite concerned about the composition of the cleaning product they’re using.
Substitution can lead to inputs that are not ideally fit for wine production. Common examples are surfactants which are not completely free-rinsing or do not have hydrotroping properties; chelates and water sequestrants that are not in formulated products or at an insufficient quantity to be effective; and silicates which are film-forming.
Sodium metasilicate (di-sodium metasilicate) appears in some laundry detergents because of the low foaming and lubricity properties theoretically assisting with soil anti re-deposition (no surface charge on garment fibre) silica coating.
Worryingly, sodium metasilicate has been transferred to wine hygiene products offered by other companies. It’s cheap and with a pH-12 it is on the border-line for dangerous goods (Technically they are Classified as a DG. Silicates can be mixed with other high pH chemicals to maintain a high aggregate pH, or, be used in a greater proportion to reduce the pH of alkali hydroxides pH 13-14).
The key reason that silicates are not fit for wine production is because they create a surface layer after cleaning. Each time the tank is washed a little more silicate is deposited and over time, it enables chemical films to form on the surfaces of tanks. These films often contain residues of alkali-denatured wine soils which, within and behind the film, yeast, microbes and other bacteria reside in the tank establishing a great risk to the overall quality of the wine.
A low to very low price for hygiene products may mean you are purchasing latent problems, or paying a lot more than you should for non-active substances or carriers such as water.
Wine Australia Annual Report
http://www.wineaustralia.com/en/About%20Us/~/media/0000Industry%20Site/Documents/About%20Us/Wine%20Australia%20Annual%20Report%202012-13.ashx
Halogens in Hygiene - Is the taint risk worth it?
Chlorine is a dirty word in wine. The family of chemically related elements to which chlorine belongs are called halogens. Halogens are toxic, they’re dangerous and they’ll taint.
There has been a significant push from within the wine community to remove halogens from the cellar and there has been an overwhelming amount of research undertaken to define sources of taint. A key concern with halogens is that a little, really does go a long way. Here is what The Australian Food and Grocery Council (AFGC) said:
“Numerous cases of contamination have been reported in the literature with “disinfectant” or “medicinal” descriptors. The problem is generally traced back to the presence of halophenols in very small amounts. These compounds have low sensory thresholds and can impart taints to foods at concentrations in the ppb (parts per billion) or ppt (parts per trillion) range.”(AFGC, 2007).
Not a lot is needed to have a big impact. Unfortunately, there is, some nostalgia around halogens. Bottling managers, Cellar Hands and even some Wine Makers have been known to talk fondly about “hypo” (short for hypochlorite). A surprising amount of wine producers will probably still have some in the cellar, which they’ll swear by. We know from our customers that there is more awareness of hygiene in the industry and many are taking some really positive, progressive steps towards their hygiene (using non-dangerous, environmentally friendly, safe, cleaning and sanitation chemicals). A minority of producers are reverting back to old habits and using a bit of “hypo” as we’re told from time to time that “nothing else works”.
And in part they’re right, halogens are incredible sanitisers. They’ve been historically used in cleaning and sanitising chemicals for all industries - dairy, household, a myriad of food and beverage production environments - for this very reason. Manufacturers are still including halogens in their products because they're a cost effective (cheap) means of ensuring exactly what it states on the packet - a clean and sanitised surface. What isn’t marketed so obviously is taint, and the large warning sign about environmental degradation caused by halogens is often omitted.
Taint is a real problem in all food processing industries.
“…the food industry worldwide has been confronted with severe tainting of various foods due to the presence of halogenated phenols (or halophenols) and their related anisoles (together referred to as organohalogens)”. (AFGC, 2007).
When looking at TCA taint caused by halogens, there is more than just the cork that needs to be taken into consideration. An article in The Australian Journal of Grape and Wine Research stated:
“TCA can originate from 2,4,6-trichlorophenol (TCP) produced from naturally-occurring phenol and chlorine from sanitisers and cleaning products, and town water.” (Simpson et al, 2007).
The potential for taint coming from multiple sources, including cleaning and sanitising chemicals is very real. We know that only a minute amount of a halogen like chlorine is needed to cause dramatic consequences.
The sensitivity of wine to this sort of taint is extreme. We’ve heard of taint caused by; residues, contaminated oak through vapor, water quality, insufficient rinsing, over-spray and we are sure there are many other methods. The chances of wine coming into contact with a halogen if it is in use in the cellar is a reality.
“Make certain that any chlorophenol-based algaecides or disinfectants used in the factory cannot be accidentally introduced into the processing line. A critical step to avoid is cleaning with water containing phenol and chlorinated cleaning agents.” (AFGC, 2007).
This is good advice, however with the multiple ways in which wines can become tainted it is almost impossible to eliminate this risk if halogen based chemicals are used in cleaning and sanitation. Wine producers are often the first to admit that accidents do happen, often during busy periods like vintage. The only way to completely eliminate this is to remove it from site.
The environment will thank you for it too, as will your staff. Halogens in the environment are toxic to many crops and plants and impact the build up of sodic soils. This sums it up quite well:
“…high tissue Cl concentrations are toxic to many crop plants, and restrict agriculture…” (White et al, 2001).
Removing halogen based products like “hypo” from the hygiene arsenal and replacing with non-dangerous, environmentally friendly products makes work sites much safer.
Halogens are a slippery slope (just look at the dairy industry) and reverting to old habits regarding hygiene isn’t the silver bullet wine producers might be hoping for. No one will disagree with wine producers wanting “hypo” like results. The reality is though, taking a halogenated approach to hygiene is an old fashioned approach that could have nasty consequences.
Looking for a non-dangerous, environmentally friendly approach to your hygiene? Then talk to us info@airdchemistry.com.
References:
Organohalogen Taints in Food, Australian Food and Grocery Council (AFGC) 2007
Philip J. White, Martin R. Broadley, 2001, Chloride in Soils and its Uptake and Movement within the Plant: A Review, Annals of Botany, Volume 88, Issue 6, Pp. 967-988.
SIMPSON R and SEFTON M, 2007, Origin and fate of 2,4,6-trichloroanisole in cork bark and wine corks, pp106–116, Australian Journal of Grape and Wine Research 13
Uhlich et al, “Analyses of the Red-Dry-Rough Phenotype of an Escherichia coli O157:H7 Strain and Its Role in Biofilm Formation and Resistance to Antibacterial Agents”
Appl Environ Microbiol. Apr 2006; 72(4): 2564–2572.
HIGH PH CLEANING IS NOT EFFECTIVE FOR BOTTLING HALLS
Caustic does not clean. And it definitely doesn't sanitise.
That brown tinge in your bottling tanks or throughout your bottling line could be causing you more problems than you think. It is a signal, and a very important one, that your line isn’t clean. Of most concern is that if it isn’t clean in areas you can see, just think about all the areas in the line that you can’t see.
The brownish tinge is often a bio-film and is putting each wine moving through your bottling hall at risk. This risk can be completely eliminated through good hygiene practices. The first step is to understand the nature of wine arriving at the bottling hall and the reactions that high pH products can have in bottling lines.
Finished wine that is about to be bottled is different to that in the cellar. These wines don’t contain heavy tartrates or high soil loads. The lighter tartrate and organic soil loading means less reliance on pH to remove wine films and surface staining. There is a tendency though, in bottling halls, to reach for high pH commodities such as sodium or potassium hydroxide (caustic) and this is where the trouble starts.
There are myths about caustic products. The most common one is that sodium or potassium hydroxide will sanitise. This is not the case. A recent study of comparative sanitising agents found that sodium hydroxide (NaOH) didn’t meet the same standards of proven sanitisers such as peroxy-acids (Abban et al, 2013). The alkaline environment does provide a small amount of resistance to microbes, yeast and bacteria, however caustic doesn’t meet the standards necessary for a surface to be deemed sanitised.
We’ve spoken about caustic products previously (read more here) and the issues that high pH products cause. There is no doubt that inadequate cleaning through the use of commodity caustic leads to protein char (or protein staining). This protein char is a biofilm and can put your wine in danger right before it enters the bottle.
“…biofilm formation in the winery is clearly undesirable as it can become a serious source of contamination.” (Joseph et al, 2007).
Biofilms are all around us (for example plaque on our teeth). When bacteria aggregate on a surface they form an extracellular glue-like matrix that protects them and attaches them irreversibly to that surface. At this point the resultant community is referred to as a bio-film (Joseph et al, 2007). The issue continues because micro-organisms living in biofilms are much more resistant to disinfectants (Vlkova et al, 2008). The really dire news is that once a biofilm has formed on the surface it is much harder to remove - you’ll need more chemical, more washes, more manual action and more time. Just more of everything to remove it.
“Biofilms are formed by different species of micro-organism are dangerous because they protect one another during the application of chemical agents (for example alkaline chlorine solutions).” (Vlkova et al, 2008)
There are more risks to high pH. Chemical manufacturers often use Sodium Metasilicate or Disodium Metasilicate to eliminate foam. At a high pH it is difficult to disperse foam in a cost effective manner. A cheap way of doing this is to use silicates. This is an issue as silicates compound layers over time to form a chemical film. This chemical film enables another environment where microbes, yeast and bacteria can persist. This effectively is stinging bottling mangers twice,firstly they have a biofilm from the caustic and secondly it is much more difficult to remove because of the silicates.
So there are some issues with caustic. Most bottling managers will state that a steam or hot water sterilisation will resolve this. On first glance this seems reasonable, anything not caught by the caustic will be eliminated through hot water or steam sterilisation. Easy right?
Well no, not really. It is extremely difficult to sterilise every part of the bottling line - from filler tanks to fittings. Some parts of the line, which may be hard to reach are simply not reaching the required temperature to create a sterile environment. This gap is then compounded by potential biofilms or chemical films protecting microbes, yeast and bacteria in the bottling hall.
“Proper cleaning enhances the effectiveness of the sanitation process. Cleaning removes the soils and enables a surface to be sanitised.” (Bar-Am et al, 2012).
The good news is that there are several steps bottling halls can take to eliminate their risk. The first is stop using caustic. Find a mid pH alkaline cleaning agent which will help wash off any light films that are produced. Make sure that the chemical does not contain any sodium metasilicate or disodium metasilicate. In our range we would suggest Destainex LF, Cleanskin or Citsanex.
In Australia and New Zealand, bottling organisations should also use a chemical sanitiser (in our range Linvasan, PerCitra or Vinisan) as a second precautionary step prior to a steam or hot water sterilisation. In the USA and Canada, Destainex LF will provide an excellent clean surface and will reduce the microbe, yeast and bacteria count significantly (through oxidation). This way you’ll be sure that your process is robust and no-hard to reach areas are going to put your wine at risk.
The benefits of moving away from caustic are immense. Not only will your bottling hall be safer and cleaner, but you’ll also reduce the amount of salt going into your waste water stream and you’ll be giving your wines far better protection.
BIBLIOGRAPHY:
Abban, Jakobsen and Jespersen, A practical evaluation of detergent and disinfectant solutions on cargo container surfaces for bacteria inactivation efficacy and effect on material corrosion, 2013, Department of Food Science, University of Copenhagen, DK -1958 Frederiksberg C,Denmark Accepted 5 June, African Journal of Biotechnology Vol.12(23) , pp. 3689-3698
Calanit Bar-Am, Jim Lapsley, Rolf A.E. Mueller and Daniel A. Sumner, Grapevines of Innovation: Ozone as a Cleaning Agent in the California Wine Industry, Journal of Wine Economics, Volume 7, Issue 01, May 2012, pp 108-125
Filomena L. Duarte, Alberto López, M. Filomena Alemão, Rodrigo Santos, Sara Canas, 2011, COMMERCIAL SANITIZERS EFFICACY – A WINERY TRIAL pg 45-52, Instituto Nacional de Recursos Biológicos
VLKOVÁ, BABÁK, SEYDLOVÁ, PAVLÍK and Jarmila SCHLEGELOVÁ, 2008
Biofilms and Hygiene on Dairy Farms and in the Dairy Industry: Sanitation Chemical Products and their Effectiveness on Biofilms – a Review.,Czech J. Food Sci. ,26: 309–323
L. Joseph, G. Kumar, E. Su, and L. Bisson, 2007, Adhesion and biofilm production by wine isolates of Brettanomyces bruxellensis, In: American Journal of Enology and Viticulture 58(3):373-378.2007
FURTHER READING
http://wineserver.ucdavis.edu/pdf/attachment/118%20sanitation%20agents%20to%20fight%20biofilms.pdf
http://academicjournals.org/article/article1380711237_Abban%20et%20al.pdf
http://www.agriculturejournals.cz/publicFiles/02507.pdf
Save Water, Recycle Your Cleanskin
There is an industry wide push to limit the environmental impact of wine production and conserve water where possible. One easy innovation that all elements of wine production can implement is re-using cleaning solutions.
Simple right. Cleaning solutions are re-used. This saves; water, chemical and time (as the solution is already mixed). The best part is that re-using cleaning chemical is cheap and can be implemented easily at any winery. The GWRDC outlined in its report that re-cycling and re-using chemical saves on water and also limits the amount of salt put into the environment (GWRDC, Operational Guidelines 2011).
The boffin’s at AIRD have put together some answers to common questions regarding how to recycle cleaning solutions.
What sort of cleaning products can be re-cycled?
As a general rule alkali cleaning products can be re-used (however we would always recommend checking with the manufacturer first). The benefit of re-using cleaning chemicals also means that you exhaust all the pH from your alkali products. This is important because it means less salt will end up in your waste water (read more here) and you’ll be saving water.
When is it ok to re-use Cleaning Solutions?
Whenever the acid levels and soil level in the vessel to be cleaned is low. Traditionally finished wine and wines with light tartrate loading don’t need a lot of cleaning action in order to remove surface soils. Often in these cases all that is required is to remove colour or films. The very light acid level (precipitated tartar) means that a high pH isn’t required.
The following are often areas where cleaning solutions can be recycled:
- Bottling lines
- Wine Tankers
- Finished Wines
- Wines with Light Tartar levels
For AIRD products if the pH of the solution is above 9 then the solution is able to be re-used.
How are other wineries recycling cleaning chemical?
A few labels on Intermediate Bulk Container (IBCs) can go a long way to ensure that effective cleaning solution is exhausted prior to pumping into the waste water stream. IBCs have been found to be effective because they are easily transported to where they are needed in the winery, or they can be placed in a warmer part of the winery (ie out in the sun) to maximise the effectiveness of the chemical solution.
Having small amounts of cleaning product is a really cheap and effective way to manage your solutions. Being able to visibly see the quality of the solution is a good control measure also as a solution with too much soil and acid in it will need to be dumped.
Will a spent solution mean a greater bacterial loading on tanks or vessels?
The microbiological loading in tanks won’t significantly change from cleaning agents. This is because the action of cleaning is the process of removing solids and soils from the surface.
Although microbes, yeast and bacteria don’t persist as freely in alkali environments (and often cleaning agents are alkali) a cleaning agent on it’s own will not necessarily impact the microbiological load.
Sanitation is what is required to reduce the microbiological loading on a surface (after it has been cleaned). A good rule of thumb is cleaning agents will address all that can be seen, sanitation will address everything that cannot be seen by the naked eye. Read more about sanitation here.
Will a higher pH mean that my surfaces are cleaner?
No. High pH doesn’t mean that your surfaces will be cleaner. The pH doesn’t equate to cleanliness and unfortunately pH alone will not sanitise either.
The reason that high pH products are used in wine production is to remove the tartaric acid which forms during the wine making process. However on surfaces that have very low levels of tartar the high pH can be a hindrance as it can cause protein char (or protein staining) which can form biofilms enabling microbes, yeast and bacteria to persist even in high pH environments (read more about biofilms here).
The pH is important regarding removing tartrates and there will become a point where the cleaning solution is “spent”, for AIRD products this is usually around a pH of 9.
If I have a higher pH does this mean that the solution will last longer and be more efficient?
No. Increasing the pH of a cleaning chemical to be re-cycled doesn’t mean that the solution is going to be more effective. In fact it is important not to overdose the cleaning product either. If a cleaning agent is over dosed, there can be issues with solubility and it also means that there will be increased salt in the waste water stream (due to more chemical). An increased dosage doesn't make the cleaning solution more effective either. This is due to the water having a carrying capacity. With a higher concentration of cleaning agent the water in the solution has a reduced capacity to dissolve acids and water plays an important role in carrying soils and solids from the tank.
The high pH is only useful to remove tartrates. The cleaning product has other ingredients such as surfactants, chelates and wetting agents which are all important to wash solids and soils in order to clean the surface. It is important to check the manufacturers guidelines about recommended dosages for a cleaning product.
Does water quality impact the dosing I should use for my cleaning solution?
Yes. Make sure that you are dosing your chemical appropriately following the manufacturers instructions. Water quality and temperature both impact upon the effectiveness of the cleaning agents that are being used (read more here). Water that is higher minerals (calcium or sodium) will need more cleaning agent. Better quality water will need less. Likewise hot water won’t require as much cleaning agent as cold in order to achieve the same result.
Why do I need to check the pH of a solution?
To re-cycle effectively you’ll need to know the pH of the solution that you are using. Having pH meters or pH test kits readily available means that you’ll be able to quickly assess the effectiveness of the chemical that you’re using. As a general rule we suggest re-using chemical if the pH of the solution is 9 or greater.
If the pH is 9 is the cleaning solution still working?
Yes. The pH of a cleaning product is not the only factor needed to clean a surface. Products that contain high quality chelates, wetting agents and surfactants are built to help remove soils and solids from the surface being cleaned and washed from the tank. The pH of a solution only becomes important when there is more acid on the surface of the vessel to be cleaned.
Every product in the AIRD cleaning range can be recycled to improve your waste water and make your cleaning spend stretch further. Now that's cleaning smarter.
The Sodastream for winemaking...
The inventors of this device claim that it will revolutionise the winemaking industry. Called the 'Miracle Machine' this contraption literally turns water into wine (with a few additions), in around 3 days.
The inventors of this device claim that it will revolutionise the winemaking industry. Called the 'Miracle Machine' this contraption literally turns water into wine (with a few additions), in around 3 days.
We would probably argue that it never will replace winemaking as a profession, just like Nespresso hasn't caused a sudden loss of jobs in the barista market.
Check out the full article: The Miracle Machine uses the magic of science to turn water into wine for £1.20 a bottle
The inventors are looking at crowd funding this project so you could be one of the first miracle makers in your village!
Managing Salt In Winery Waste Water
Effectively managing salt levels in winery effluent is a significant issue for wine production. The use of sodium in particular through caustic soda has significant environmental impact which can lead to the build up of sodic soils.
Effectively managing salt levels in winery effluent is a significant issue for wine production. The use of sodium in particular through caustic soda has significant environmental impact which can lead to the build up of sodic soils.
To produce the perfect drop there is a lot of water, unspent grapes, juice, alcohol, sugar and a raft of other chemicals that will eventually end up flushed into the waste water stream. Leading industry bodies all argue that taking action early and limiting the amount of contaminants that get into your waste water not only is commercially savvy but will do your waste water a world of good too.
In wineries, wastewater mostly comes from cleaning operations and is “…primarily made up of wine, grape juice suspended solids (during vintage) and cleaning agents (eg NaOH, KOH)” (Mosse et al, 2011).
The inputs that are washed into the waste water stream can have a significant environmental impact and Mosse argues that it is essential that wine producers know the composition of their waste water in order for improvements to be made. The “… minimisation of both quantity and content of solutes and solids is desirable on both economic and environmental fronts” (Mosse et al, 2011).
In wineries salts form as a result of a reaction between strong alkali cleaning agents and wine. This has a significant environmental impact on soil and water quality, especially if high pH cleaning agents (eg caustic) are used.
The issue with too much sodium in the environment is that it reduces the availability of water to plants (Rengasamy et al,1994) and creates a saline environment. Mosse argues that a highly saline environment combined with high levels of organic matter (another by-product of wine production) exacerbate the clogging of soil pores and in turn further limit plant growth (Mosse et al, 2011). This has the potential to also lead onto sodic soils.
The Australian Wine Industry Public Environment Report (2003) indicated a value of 0.812 kg caustic/tonne of grapes (Kumar et al, 2009). For a winery that crushes 500 tonnes this means that over 400kg of caustic cleaning agent will typically be washed into their waste water stream annually.
What this all equates to is a significant amount of salt being produced and then flushed out into your winery soil. Once the salt has been flushed into the waste water stream there are not many feasible means for wineries to remove it. Highlighting the argument for preventing too much salt from entering the waste water stream.
Salt removal processes such as reverse osmosis are often expensive and require specialised equipment/infrastructure. A further problem with salt removal technologies is that a highly concentrated brine is often produced, which requires specialised disposal (Metcalf and Eddy 2004)(Mosse et al, 2011).
To counter issues associated with sodium, many winemakers are looking to potassium based cleaning agents. While there are associated benefits to soil fertility with potassium, Mosse (2011) states that long term application may still lead to changes in the physiochemical properties of the soil structure. The GWRDC makes it very clear that:
“Salts and many chemicals cannot be cheaply removed in treatment. Keeping them out of effluents to begin with, by reducing their use or early segregation, can be very effective.” (Day et al, 2011).
There is no doubt that by using less chemical there will be less salt washed into the waste water stream. However when looking at reducing salts in waste water, another key concept to take into account is the pH of cleaning products. Higher pH cleaning agents tend to have a faster rate of reaction, which in the modern winery is a valuable commodity. However, the downside is that they create more salt as a by-product, which ultimately ends up in the waste water stream. This is because a greater concentration of OH- ions are required to be neutralised from higher pH solutions. However, we know from basic chemistry that the rate of reaction can also be increased by using hot water (40-60 ºC). So rather than increasing the concentration of your cleaning products to increase pH, hot water can be used as an effective means to increase cleaning efficiency and reduce salt by-product output.
Every winemaker will tell you, the most tenacious soil to remove from a tank is tartrate. The reality is that tartrates can be removed with a non-dangerous, zero salt producing cleaning weapon. Warm water. The fact is, that if you are using chemicals to clean your tartrates, you need to ensure there is sufficient water to enable the tartrates to be dissolved in solution and thus removed from your tank walls. Chemicals only assist the water in doing it’s job, they don’t replace it.
AIRD alkaline cleaning products have a lower pH, to minimise salt production, and have additional surfactants to keep organic solids in solution meaning you can wash everything from your tanks. Give them a go - you’ll be glad you did!
Further Reading:
References
Day et al, 2011, Winery Waste Water Management and Recycling Operational Guidelines, Grape and Wine Research and Development Corporation
Kumar A, Frost P, Correll R, Oemcke D (2009) Winery wastewater generation, treatment and disposal: A survey of Australian practice. CSIRO. Adelaide SA.
Mosse et al, 2011, Review: Winery Waste Water Quality and Treatment Options in Australia, Australian Society for Viticulture and Oenology Inc, Australian Journal of Grape and Wine Research 17 p111-122.
Rengasamy et al, 1994, Introduction to Soil Sodicity, Co-Operative Research Centre for Soil and Land Management, University of Adelaide in conjunction with the CSIRO
Produce Better Wine - Clean and Sanitise Your Winery!
Cleaning and sanitation is important because it enables wine producers to control what comes into contact with their wine. There is overwhelming evidence that states cleaning and sanitation in wine production significantly reduces the risk of taint by yeast, microbes and bacteria. Two basic processes are cleaning and sanitising and we have outlined a quick re-cap of both processes below.
We would say that. But we’re not the only ones.
Dharmadhikari argues that “Cleaning and sanitation is crucial to producing quality wine” (Dharmadhikari, 1999).
Oelofse states “…general poor cellar hygiene along with improper cleaning and sanitisation of barrels – a critical source of Brettanomyces/ Dekkera contamination of wine…” (Oelofese, 2008).
Rankine makes a very valid point when he presented that “…the presence of contamination, whether microbiological or otherwise, during the making and maturation of wine can result in a lowering of quality” (Rankine, 2004).
He went on to state that consumers have a right to expect wine “…is made from sound grapes in a clean winery” (Rankine, 2004).
Fair enough. So it’s clearly not just us at AIRD. Cleaning and sanitation is important because it enables wine producers to control what comes into contact with their wine. There is overwhelming evidence that states cleaning and sanitation in wine production significantly reduces the risk of taint by yeast, microbes and bacteria. Two basic processes are cleaning and sanitising and we have outlined a quick re-cap of both processes below.
“Cleaning is the complete removal of food soils/solids using appropriate detergent chemical under recommended conditions” (Schmidt, 2009).
Most cleaning in wineries is CIP (clean in place) and involves a circulation of a solution through a spray ball. As wine is acidic, alkaline cleaning agents are used to break down tartar and other organic particles remaining in the tank. Simple enough - however there are some chemicals to be wary of when cleaning in the cellar. Products that contain silicates (sodium metasilicate or disodium metasilicate) will create a chemical film in tanks that can enable bugs to persist, and chemicals that do not contain sufficient surfactants will leave protein in the tank that can lead to a biofilm (Schmidt, 2009). Cleaning regularly ensures that you don't have bio-films in your tanks and will enable you to rest easy knowing that a risk of taint has been eliminated.
“Sanitise refers to the reduction of microorganisms to levels considered safe from a public health viewpoint.” (Schmidt, 2009)
Sanitation is different from cleaning. Sanitation takes place after cleaning and refers to the reduction of micro-organisms (it is reduction as opposed to elimination of all micro-organisms which is sterilisation).
Sanitation can be achieved through chemicals or through temperature. Thermal sanitation involves the use of hot water or steam for a specified time period and chemical sanitation involves the use of an approved chemical sanitiser at a specified concentration and contact time (Schmidt, 2009).
Chemical sanitisers usually will work through oxidation or pH and are usually acidic (stabilised peroxy-acids). For wineries the reduction in micro-organisms, yeasts and bacteria through sanitation can significantly reduce the risk of wine taint caused by bugs. Sanitisers take care of what can’t be seen - and is a further measure that wine producers can use to control what their wines come into contact with in their tanks at a microscopic level.
There are significant benefits to be had from a bit of elbow grease and common sense. Working smarter with your cleaning and sanitation protocols will ensure that you’re wine isn’t at risk of taint this vintage. Like to know more - check out our products here.
References:
A. Oelofse et al, 2008, “Significance of Brettanomyces and Dekkera during Winemaking: A Synoptic Review”, Institute for Wine Biotechnology, Stellenbosch University, Matieland (Stellenbosch), The Australian Wine Research Institute
Bryce Rankine, 2004, “Making Good Wine”, McMillian Company, pg 225
Murli Dharmadhikari, 1999, “Importance of Cleaning and Sanitation in the Winery, Vineyard and Vintage View”, Mountain Grove MO
Ronald H. Schmidt, 2009, “Basic Elements of Equipment Cleaning and Sanitizing in Food Processing and Handling Operations”, FS14 is one of a series of the Food Science and Human Nutrition Department, Florida Cooperative Extension Service, Institute of Food and Agricultural Sciences, University of Florida. Original publication date July 1997. Revised March 2009. Reviewed January 2012
Good hygiene depends on high quality water.
A final rinse after cleaning and sanitising with low-quality water that contains bacteria, or is too hard for chemical to be effective could be putting your wine at risk when it doesn’t have to be. By using high quality water in your winery operations, you can minimise contamination risk, without significant financial expenditure. There are two principal considerations to take into account to assess water quality - water hardness and potability.
A final rinse after cleaning and sanitising with low-quality water that contains bacteria, or is too hard for chemical to be effective could be putting your wine at risk when it doesn’t have to be. By using high quality water in your winery operations, you can minimise contamination risk, without significant financial expenditure. There are two principal considerations to take into account to assess water quality - water hardness and potability.
As wine is a potable good, it is common sense to make sure that the water used in a winery is to a potable standard. If you are using rain water - it is important to make sure that there is no bacteria, yeast of microbes that are persisting in your tanks as this may impact your final rinse and your overall hygiene standards. You can treat your rainwater with filters which can dramatically improve water quality. High quality filtered water is essential for bottling lines and is useful to protect finished wines. Bore water should be treated with similar filtration methods so that you can maintain the highest possible hygiene standards in the winery.
Water hardness is a measure of the mineral content in water (hard water having a high mineral content). When water hardness is being quoted, the value being talked about is the ‘total permanent hardness’ of water, which is the amount of Calcium and Magnesium sulphates in water, but is expressed as an equivalent of Calcium Carbonate (CaCO3).
Let’s look at a practical example, say the Barossa Water System in South Australia. The total permanent water hardness in this system sits at 148 mg/L of CaCO3. According to the United States Geological Survey, this puts the water in the Barossa in the ‘Hard’ classification (between 121 and 180 mg/L is classified as ‘Hard’). While water hardness is not detrimental to health, it is definitely a negative in an industrial setting.
Firstly, the hard water is more corrosive, especially in galvanic corrosion. So using hard water decreases the useable life of your stainless components. Don’t forget that hard water also deposits scale on metals, which can clog up your pipes too. More importantly, hard water also reduces the effectiveness of your cleaning and sanitising chemicals.
Hard water has many free 2+ ions that combine with metal salts in hygiene chemicals to form soap scum. Essentially the minerals in the water reduce the surfactant properties of your cleaning and sanitising agents. This in turn reduces the rinsing properties of your solution. The surfactants role in cleaning agents is to keep surface tension in solution high, which keeps insoluble molecules suspended in solution so they can be washed away. If the reduction in surfactant properties is high enough, you will be left with deposits on your tank walls which will require further work (costing you time and money).
To combat hard water more chemical may be required. The use of additional chemical should be carefully considered, as there will be a flow on impact to your effluent. If water hardness is impacting the cleaning efficacy of your chemical to an extent where deposits are being left on your surfaces, it would be detrimental to simply leave the deposits on your stainless surfaces, as this would provide an excellent breeding ground for microbes such as Brettanomyces.
Most good industrial cleaning products should contain ingredients to counteract the effects of hard water, however if you live in a particularly ‘hard’ water area, such as our friends in the Barossa, then water softening could be a worthwhile investment. Water softening does come at a risk of higher Sodium or Potassium deposits in your soil, as they are the two main salts used to soften water (in ion transfer softening). However you want to treat hard water, it is worthwhile knowing what your water hardness level is, and then carefully consider the impact of treatment or additional chemical on your effluent.
By using potable water, and being aware of the hardness level of your water, you offer your cleaning chemicals the best opportunity to effectively clean your surfaces. As the saying goes, rubbish in equals rubbish out. So don’t start with rubbish water quality, otherwise you’ll get rubbish wine out.
References
Aquasafe systems (2005) Quality of Mains Water Assessed Against Water Quality Criteria, http://www.aquasafecanada.com/quality-of-mains-water-assessed-against-water-quality-criteria/, Accessed 2013-12-06
Wikipedia (2013) Hard Water, http://en.wikipedia.org/wiki/Hard_water, Accessed 2013-12-06
How do you open a wine bottle without a corkscrew? You use your SHOE!
Yes, the video is in French, but you can turn the subtitles on, or watch and be amazed!
Yes, the video is in French, but you can turn the subtitles on, or watch and be amazed!
Otherwise, Mirabeau Wines owner Stephen Cronk has shown internet users how to uncork a bottle using only a shoe, a wall and a bit of muscle power.
Is peracetic acid putting your wine at risk?
Sanitation is important. We strongly recommend that every winery implements high quality sanitation protocols. Good hygiene through a fully built cleaning agent and a sanitiser (that won’t taint wine) will reduce the risk of unwanted bacteria yeast and microbes taking hold in your wine. Ultimately it protects your investment. Chemical products used in the cellar for cleaning and sanitation that taint wine increase the likelihood of wine spoilage through human error.
- Sanitation enables winemakers to control what their wine comes into contact with
- Peracetic acid sanitisers break down to hydrogen peroxide and acetic acid
- Acetic acid is one of the primary causes of VA
Peracetic acid smells like ants, breaks down into acetic acid and could result in your wine smelling like nail polish remover.
The need for sanitation has never been greater - however without the right tool for the job your wine may be suffering. Protecting your wine with a sanitiser is best practice hygiene. However the use of sanitisers that contain peracetic acid can result in an inadvertent acetic acid addition - increasing the risk of volatile acidity (VA).
Sanitation is important. We strongly recommend that every winery implements high quality sanitation protocols. Good hygiene through a fully built cleaning agent and a sanitiser (that won’t taint wine) will reduce the risk of unwanted bacteria yeast and microbes taking hold in your wine. Ultimately it protects your investment. Chemical products used in the cellar for cleaning and sanitation that taint wine increase the likelihood of wine spoilage through human error.
The wine industry needs skilled labour to produce wine. This reliance on staff means there is an inherent risk of mishaps. Mistakes are made - especially when workers become tired during busy periods like vintage. If peracetic acid sanitisers come into contact with wine the net result is an addition of acetic acid to your wine. This poses a significant problem.
Sanitation Guide
Adding acetic acid to any wine is concerning. Basic organic chemistry tells us that peracetic acid will break down into hydrogen peroxide and acetic acid (Bird, 2010 p216). The hydrogen peroxide will form water and oxygen. The acetic acid is something that winemakers are almost universally trying to prevent coming into contact with their wine. This is because of the risk of taint through VA.
One of the primary causes of VA is acetic acid (the acid of vinegar). Although acetic acid is a natural component of wine, an oversupply of acetic acid can lead to wine spoilage (Bird, 2010 p255). If a wine does have acetic acid issues this can lead to further work in filtration, reverse osmosis or having to isolate and blend the affected wine. This takes time and further increases the cost input into finished wine.
The AWRI states in their report “Wine Bacteria - Friend or Foes” that there is a high risk of spoilage caused by acetic acid bacteria during prolonged barrel maturation and poor management (of acetic acid) during bottling and storage of red wine (AWRI Wine Industry Journal, 2009). Acetic acid bacteria are also able to survive in ethanol and sugar rich environments (Bartowsky and Henschke, 2008). The use of a sanitiser during these periods reduces the risk of further unwanted bacteria, yeast and microbes coming into contact with wine. However peracetic acid based sanitisers increase the risk of inadvertently making an acetic acid addition to your wine - especially if a single pass rinse is not completed properly.
Acetic acid bacteria have been shown to contribute to volatile acidity in wine must and the production of acetic acid can contribute to sluggish or stuck ferments also (Du Toit and Lambrechts, 2002). Peracetic acid presents a risk that can be avoided easily through the use of chemicals that will not taint. This eliminates the risk of human error in the cellar and gives winemakers some assurance that spoilage through VA will not be caused through chemicals used on site.
We don’t believe products that can taint wine should be utilised in wine production. As such we would recommend any of our sanitisers (which can be viewed here), Destainex or Destainex LF will provide effective sanitising and will not taint your wine. Give them a go - your wine will thank you for it!
References
Bartowsky, E.J. and Henschke, P.A. (2008) "ACETIC ACID BACTERIA SPOILAGE OF BOTTLED RED WINE - A REVIEW",International Journal of Food Microbiology 125, 60-70.
Bird D, (2010), “UNDERSTANDING WINE TECHNOLOGY” third edition, p72, p138, p139
Du Toit, W.J. et al (2002) "THE ENUMERATION AND IDENTIFICATION OF ACETIC ACID BACTERIA FROM SOUTH AFRICAN RED WINE FERMENTATION'", In International Journal of Food Microbiology, vol. 74, 2002, p. 57– 64.
Eveline J. Bartowsky,et al (2009), “WINE BACTERIA - FRIEND OF FOES” - AWRI Report, March/April 2009, Vol 24 NO 2, Wine Industry Journal
Kantor et al (2013)“LACTIC ACID AND ACETIC ACID BACTERIA ISOLATED FROM RED WINE”, Journal of Microbiology, Biotechnology and Food Sciences (special edition) pp1704-1715
Links
http://www.newworldwinemaker.com/pdf/AWRI_report_wine_bacteria_friend_or_foe.pdf
http://www.jmbfs.org/wp-content/uploads/2013/06/69_jmbs_kantor_fbp_m.pdf
The real cost of cleaning
Conducting a strategic review of winery processes and procedures can lead to significant gains in efficiencies and reduce labour costs.
Conducting a strategic review of winery processes and procedures can lead to significant gains in efficiencies and reduce labour costs.
All wineries need to clean. Cleaning (and sanitising) ensures that winemakers can control what goes into wine and what doesn’t. This inevitably means that wineries will have to spend money on hygiene products. Most will do what they did last year, or the year before. While this may seem like an attractive proposition, it does pay off in the long-term to think strategically about your hygiene needs, and make purchasing decisions with those strategic requirements in mind.
To be strategic we need to take a holistic view of the cost inputs with our cleaning and hygiene procedures. Many cost comparisons of hygiene products only consider the chemical cost, and don’t take into account all the real costs and opportunity costs associated with cleaning. Chemical cost is a part of the real cost of winery hygiene, but so is labour cost, efficacy costs (having to conduct repeat processes) and additional process requirements (think citric rinses after caustic washes). The opportunity costs, while hard to evaluate and predict, can be devastating if not accounted for appropriately. They include environmental impact, safety and spoilage risks. The view that chemical cost is the only important factor in winery hygiene costings is very simplistic, and it could be doing damage to your bottom line.
Real Costs
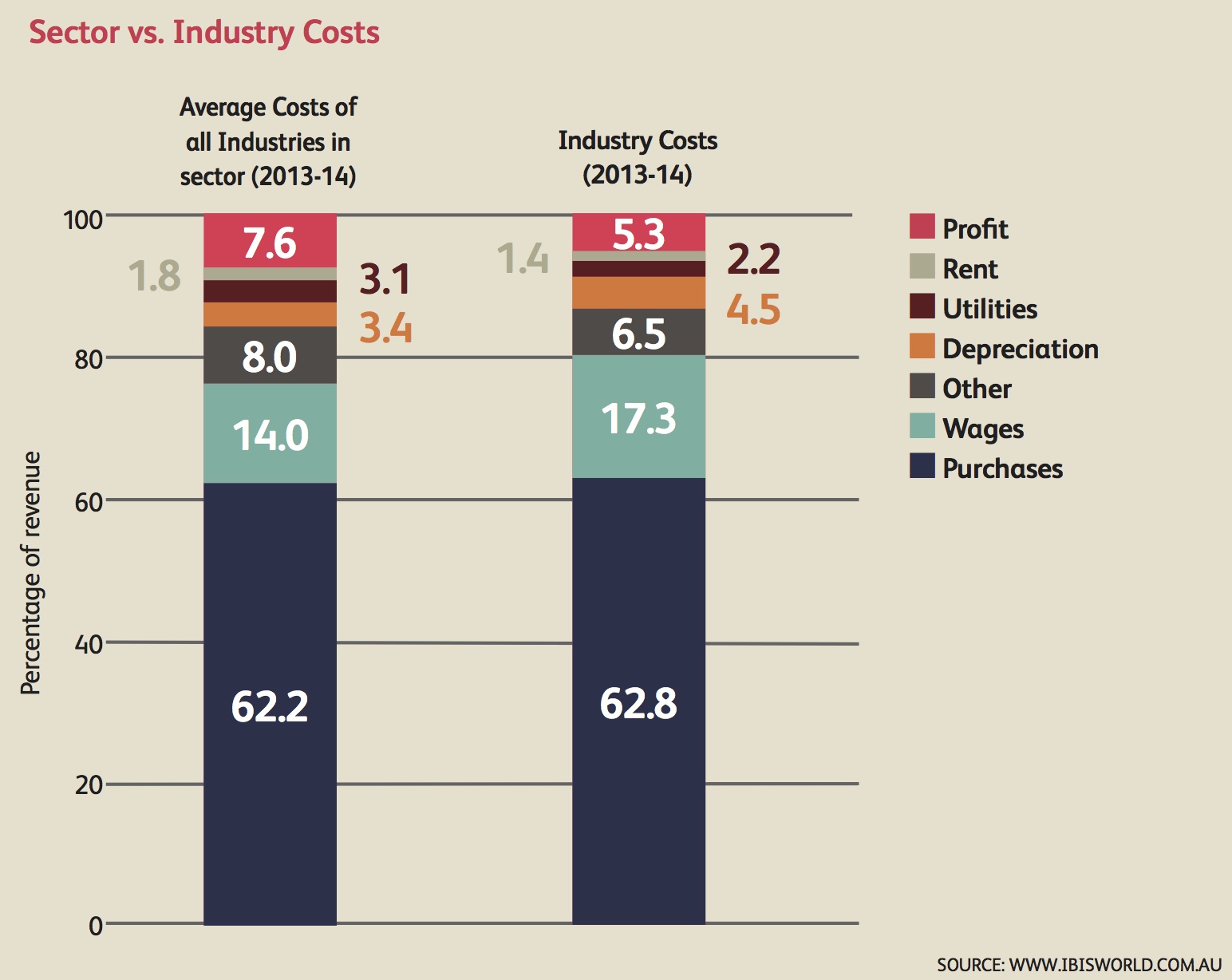
Let’s start with labour, which according to the November 2013 IBISWorld wine report, accounts for 17.3% of revenue in wine production (Lin 2013, p.24). At almost a fifth of the industry average total revenue generated, this equates to a significant amount of money spent on labour in wine production. Furthermore for every $1.00 that is paid as a labour expense a further $0.26 is required to be spent on capital investments (Lin 2013, p.35). What we see here is that the wine industry on average, spends 3.3% more of total revenue on labour costs compared to all other industries (Lin 2013, p.24). The take home message is that reducing labour costs can increase your annual revenue intake and the modern winery should be seeking out efficiency improvements to reduce their labour overheads.
Figure 1: Wine sector costs vs Industry Costs (Lin 2013, p.24)
Another significant winery hygiene cost is chemical. This cost is harder to quantify as tank sizes vary, tartrate levels change and there is volatility in the price of chemicals, quality and the quantity. Water is another cost in winery hygiene, and while in comparison to labour and chemical costs, it is much smaller, any reduction in water usage is a cost saving and is environmentally more efficient.
Let’s take a look at one of the more popular cleaning agents used in wineries, sodium hydroxide (or caustic soda) and what it actually costs to clean a tank. Most wineries will undertake the following process to clean a tank when cleaning with sodium hydroxide:
- Single pass water rinse
- Sodium hydroxide circulation
- Single pass water rinse
- Citric acid circulation
- Single pass water rinse
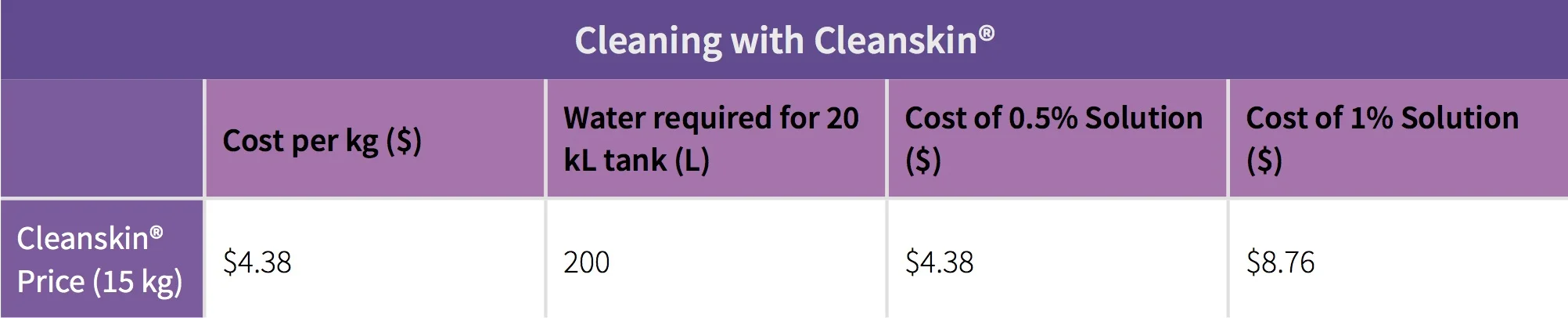
The standard procedure for cleaning with sodium hydroxide is use a 1-2% w/v aqueous solution first, and then perform a citric acid rinse of 0.5-1% w/v (to reduce the pH level of any residual chemical). Sodium hydroxide comes in two forms - as a liquid or in a solid pearl form. When reviewing pricing the cheapest commodity price we could find for bulk purchases was $0.65 per litre for the liquid and in a pearl it was $1.00 per kg. Citric acid costs between $1.25-$1.50 per kg.
Chemical Input Costs
Table 1: Sodium Hydroxide and Citric Acid chemical costs per wash.
The total chemical input cost for the liquid price is almost $6 per tank and the average price is $8.50 for a 20kL tank. If it were only this cheap to clean a tank! The issue is that staff are required to set the pumps and equipment up in order to achieve this and this is where the exercise becomes expensive.
Labour Costs
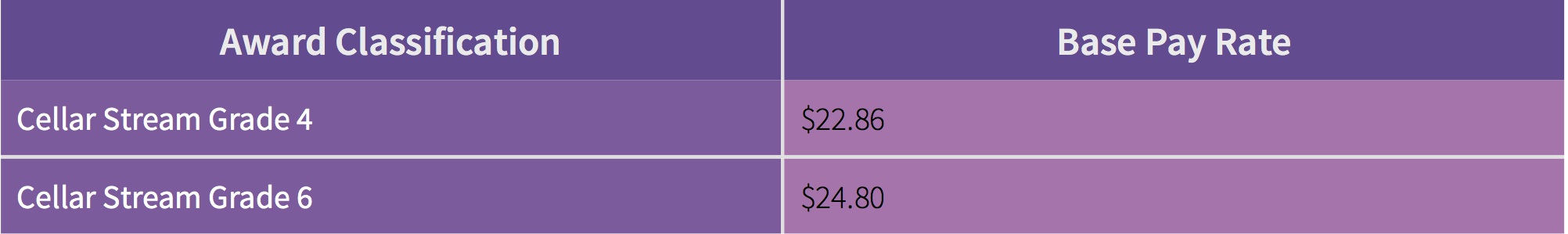
Table 2: Wine Industry Award Wages (FairWork Ombudsmen 2012, pp.4-9)
If we presume it takes 30-60 minutes to clean a tank, we can see that the labour cost far outweighs the chemical cost. If we take the Cellar Stream Grade 4 award then it costs $0.38 for every extra minute that is spent cleaning tanks in labour. Looking at this strategically, it makes sense to be looking at ways to minimise time that staff are spending cleaning. The more efficient they are the more of the 17% of total revenue that you’ll have back in your pockets!
Our focus at AIRD has always been on reducing processes, that is, cleaning smarter. One of the first processes that can be eliminated by using AIRD products is a citric rinse. If it takes 5-10 minutes to run a citric rinse on each tank, it is actually costing between $1.90 and $3.80 per tank in additional labour expenditure. Over the course of a vintage this adds up significantly (just think how many times your tanks are cleaned in a vintage). None of our products require a citric rinse, which reduces your cost input for each wash.
Eliminating a citric rinse also eliminates the need for a water rinse after the citric also - and if it takes 2-5 mins for a single pass water rinse then there is a cost of $1.14 - $1.90 which can be saved. If you were looking to compare the process for Aird cleaning chemicals including Cleanskin® the process is as follows:
- Single pass water rinse
- Cleanskin circulation
- Single pass water rinse
So a five step process has been reduced to a three step process - with better chemical inputs. Labour processes have been reduced significantly with Cleanskin® saving between $3.04 - $5.70 per tank in labour costs. Streamlining processes is not only working efficiently but it reduces labour costs significantly.
More efficient processes and better inputs reduce the likelihood of tanks requiring a repeat clean. There is a high cost for every time a tank needs a second clean with sodium hydroxide because five steps are necessary to work through in order to clean. Sodium hydroxide as a commodity will also leave a protein stain in tanks. An overwhelming majority of wineries we have spoken to in relation to protein staining will either treat protein stains with additional chemical or get staff to get in and scrub tanks where they can reach. For each tank labour costs to re-clean tanks are $0.38 per minute plus additional chemical required.
There are savings to be made in reducing processes and making cleaning a more efficient process. Investing in better inputs means that less time is spent repeat cleaning and without the need for old fashioned citric rinses.
Product Comparison
Still not convinced? Lets look at what it would cost to clean a tank with Cleanskin®:
Table 3: Cleanskin® chemical cost per wash
When you include the labour time to undertake a citric and water rinse it becomes extremely close regarding input costs. The additional processes of a 5-10 minute citric rinse and a 3-5 minute water rinse required for cleaning with sodium hydroxide are costing you an additional $0.18 per tank when compared to Cleanskin®. If you are paying more than $0.65 per L for your sodium hydroxide it becomes much more expensive.
Table 4: Caustic/Citric overall cost per wash
Table 5: Cleanskin® overall cost per wash
So not only is Cleanskin® cheaper, but the other benefit is that the chemical you’re using will not leave protein stains in your tanks - leaving you with less repeat cleaning to undertake. It makes staff more efficient, your hygiene much better and your approach to cleaning more strategic.
Opportunity Costs
This brings us to opportunity costs, which is essentially a cost for not doing something (oftentimes described as the cost of missing out on the benefit of not doing something). This cost does vary, and the nature of opportunity costs are somewhat speculative, however they are real, and can result in serious long-term consequences if not taken into account. It is well known that biofilming from protein stains enables yeasts, bacteria and microbes to persist in tanks. This can lead to issues such as taint, or unwanted microbe contamination of your finished product. The opportunity costs here are the loss of income due to poor wine quality, and then the necessary costs to manage or remove the unwanted microbes from your winery. In comparison, the real costs of cleaning properly are much cheaper than the opportunity costs in this scenario. Better hygiene is better protection for your wine.
That was just one example of opportunity costs in your winery. There are plenty more such as; work, health and safety and environmental management to name a few. It is important that in your strategic thinking about cleaning, you look at the opportunity costs and how they may impact upon your winery, and then make a decision weighing up all the benefits.
Conclusion
In the end, the strategic move is to know what your costs are and how they relate to the finished product. You may be surprised to learn that a premium product not only is cheaper in the long-term (through reduced labour costs), but better for your wine and therefore better for your bottom line!
References
Lin, R 2013 ‘IBISWorld Industry Report C1214: Wine Production in Australia’, IBISWorld, Nov. 2013,www.ibisworld.com.au/gosample.aspx?cid=61&rtid=101?, accessed 2013-12-03
FairWork Ombudsmen 2012 ‘Wine Industry Award: Pay and Conditions Guide’, Australian Government,http://wgcsa.com.au/wp-content/uploads/Wine-Industry-Award-Rates-and-Conditions-01-06-2012.pdf, accessed 2013-12-03
Wine opinion: why some aromas are better left out of the bouquet
"Unscrupulous winemakers still don't clean their equipment properly, and use bad fruit. So, as the saying goes, 'rubbish in, rubbish out.' But this wine still has to be sold."
"Unscrupulous winemakers still don't clean their equipment properly, and use bad fruit. So, as the saying goes, 'rubbish in, rubbish out.' But this wine still has to be sold."
You can read the whole article here. The point we like to take out of this is that while some bacteria and microbes are beneficial to your wine, others are not. We know, it's not revolutionary stuff, but the point of cleaning and hygiene is to control for the bad stuff, whilst still maintaining the good stuff.
Ancient wine cellar reveals new discovery
Calling all ancient wine aficionados! It turns out that wine appreciation is an ancient art, in fact 3,700 years old!
Calling all ancient wine aficionados! It turns out that wine appreciation is an ancient art, in fact 3,700 years old!
" Scientists have uncovered a 3,700-year-old wine cellar in the ruins of a Canaanite palace in Israel, and chemical analysis shows this is where they kept the good stuff. "
"'It's not wine that somebody is just going to come home from a hard day and kick back and drink,' said Andrew Koh of Brandeis University. He found signs of a blend of ingredients that may have included honey, mint, cedar, tree resins and cinnamon bark. "
Wine that lasts a little longer
"The researchers added chelation compounds that bind with metals to inhibit oxidation, or oxygen's ability to react with some of the trace metals that are found in the wine, according to Gal Kreitman, a doctoral candidate in food science, Penn State."
"The researchers added chelation compounds that bind with metals to inhibit oxidation, or oxygen's ability to react with some of the trace metals that are found in the wine, according to Gal Kreitman, a doctoral candidate in food science, Penn State."
If it all sounds a bit too technical your liking, then we at AIRD have a suggestion to help slow down the oxidation of your wine... Drink it before it oxidises (responsibly of course)!
You can read the full article here - it's promising research to help consumers enjoy every last drop!
Why does wine cry?
Grape crushing is essentially harvesting grape tears right? Ok, so that's probably a bit off the mark, but to understand why wine 'cries' or has 'legs' this YouTube video gives a far better explanation.
Grape crushing is essentially harvesting grape tears right? Ok, so that's probably a bit off the mark, but to understand why wine 'cries' or has 'legs' this YouTube video gives a far better explanation. It's pretty neat stuff. Also, surface tension plays a huge role in the quality of your final clean - just to tie it back to what we do best, that's why we add surfactants to our many of our cleaning products to assist with the final clean. But that's another story!